VISUAL AID TO PROCESS IMPROVEMENT, REALISTIC GOAL SETTING AND RESOURCE MANAGEMENT
This article discusses how Funnel Diagrams can be used to make a positive impact on your lead optimisation projects.
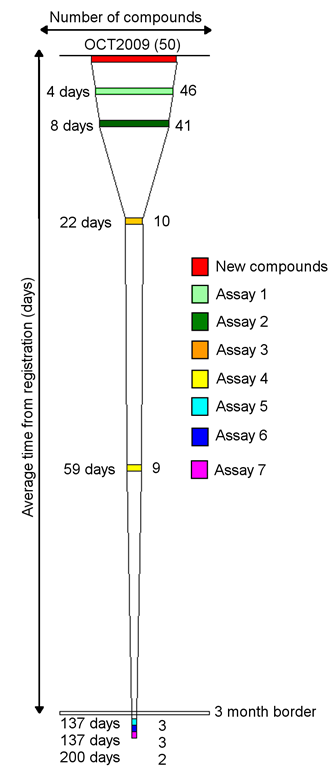
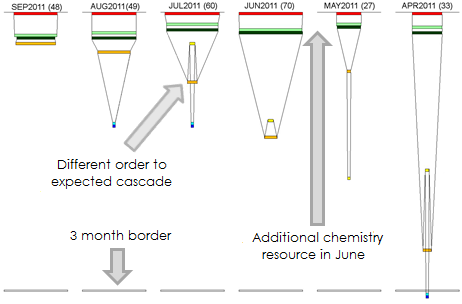
Topics include: spotting process bottlenecks, confirming the successful impact of process changes, gaining insights into how achievable a goal is, taking a glance at how resources are split across multiple projects and assessing resource needs.
Two example lead optimisation projects that are at different stages are used to illustrate the funnel applications. An error bar extension to the original visualisation is presented.
Hirons L. Chemistry Today, August 2012, 30(4), 24 – 26
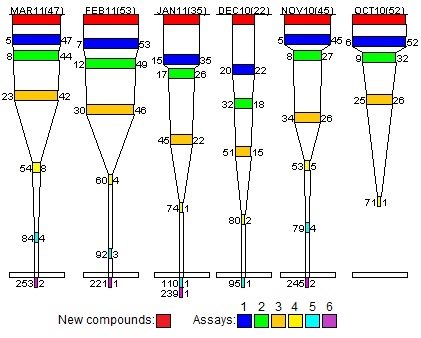
Funnel Diagram for project A:
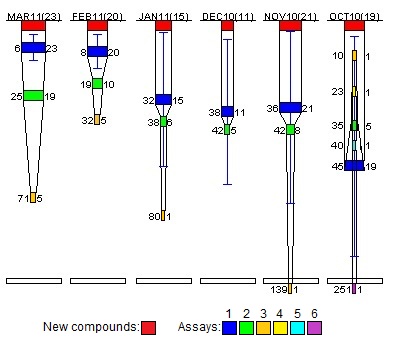
Funnel Diagram for project B with Assay 1 error bars: